Book traversal links for 3.3.1.3 Chest radiography
CXR is recommended by WHO to be used in parallel with the W4SS where CXR is available to assist in ruling out active TB prior to initiating TPT among people living with HIV who are on ART. The GDG agreed that, due to the increased sensitivity, the evidence supported using CXR in addition to the W4SS as a parallel screening strategy in which a positive or abnormal result on either screen would indicate a referral for diagnostic evaluation. Data on “any abnormality” and an “abnormality suggestive of TB” detected by CXR were reviewed and either approach is recommended, depending on the context, the availability of radiological expertise, resources and preference towards higher sensitivity or higher specificity.
The evidence reviewed for the performance of CXR and the W4SS for all people living with HIV came from 8 studies conducted in Benin, Botswana, Brazil, Guinea, India, Kenya, Malawi, Myanmar, Peru, South Africa and Zimbabwe, with a total of 6 238 participants (see Web Annex B, Table 14, and Web Annex C, Table 7). The average prevalence of TB in all people living with HIV in the studies was 7%, ranging from 3% to 18%. Among outpatients on ART, the average prevalence was 2.6%.
CXR alone was found to have similar sensitivity to and similar or higher specificity than the W4SS across all subpopulations. When combined in a sequence whereby CXR followed a positive W4SS screen, CXR had a lower or similar sensitivity with higher or similar specificity. When combined and used in parallel with the W4SS, it had a higher or similar sensitivity and similar specificity (see Table 7). The IPD meta-analysis found this strategy to have the highest sensitivity (0.85; 95% CI: 0.69–0.94) compared with the W4SS (0.53; 95% CI: 0.36–0.69) and the other tools and strategies assessed for TB screening in outpatients on ART. While the data were limited for inpatients living with HIV, the combined strategy of CXR and the W4SS had a very low specificity (0.07; 95% CI: 0.03–0.19), similar to findings for using CRP or the W4SS alone (see Web Annex C, Table 7).
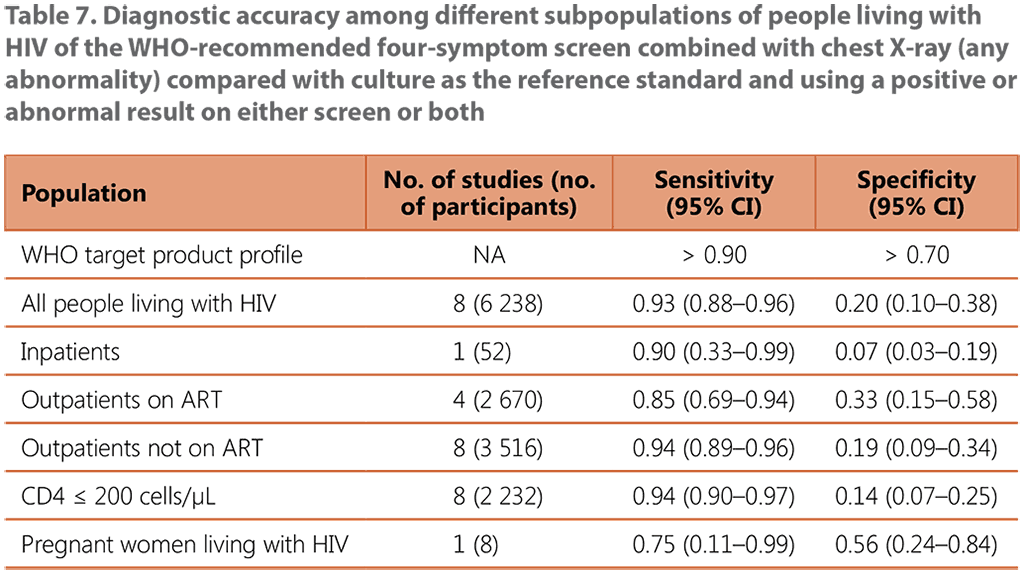
ART: antiretroviral treatment; CI: confidence interval; NA: not applicable.
 Feedback
Feedback