Book traversal links for Annex 5. Checklist for PMTPT components in reviews of national programmes
Background: A programme review is an integral part of the management cycle of a TB programme. It is a structured way of assessing the performance of the programme and for improving its quality; it is also the basis for developing or updating the national strategic plan. PMTPT is one of several preventive actions envisaged in the End TB Strategy, such as TB case-finding, infection control, prevention and care of comorbidities (e.g. HIV), access to universal health care, social protection and poverty alleviation.
Objectives: The review should determine how TPT is implemented in public and private health services, by ensuring that:
- national guidelines are updated in line with the latest global recommendations, and the national strategic plan provides adequate resources for implementation of the guidelines;
- appropriate staff, stakeholders and expert groups are engaged in implementing the guidelines;
- clear guidance is available on identifying target populations for TPT, ruling out TB disease, diagnosing TB infection or assessing patient eligibility for TPT, and providing TPT when necessary;
- medication support is available to help people start and complete TPT effectively;
- data are recorded systematically to monitor indicators in the cascade of care for TPT; and
- other implementation support is available, such as training, advocacy and management of commodities.
Various personnel play a role in TPT implementation. Staff to be interviewed include:
- managerial staff for the national and subnational TB programmes and the national HIV/ AIDS programme;
- health workers and community health workers involved in TB and HIV care, household contact evaluations, diagnostic services in health-care facilities (in both the public and private sectors and at both primary and secondary levels of health care); and
- staff in other state sectors, such as primary care, hospitals, occupational health services (e.g. mining industry), prison health services and migrant screening facilities.
Table A5.1 provides a checklist of TPT components to be used in reviews of NTPs (1). The elements should be available in their NTP, guidelines and national strategic plan (2).
Table A5.1 Checklist of TPT components for programme reviews
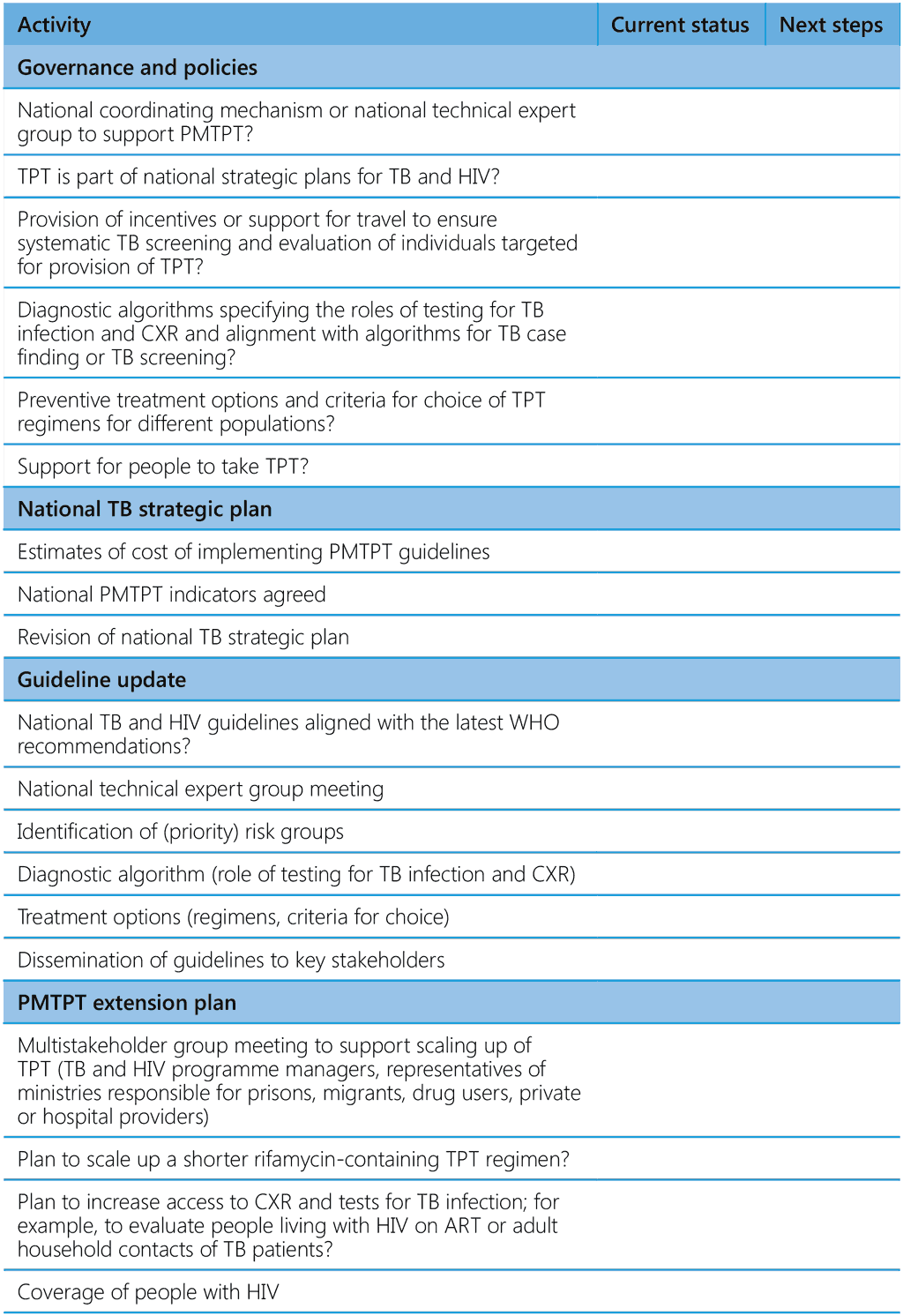
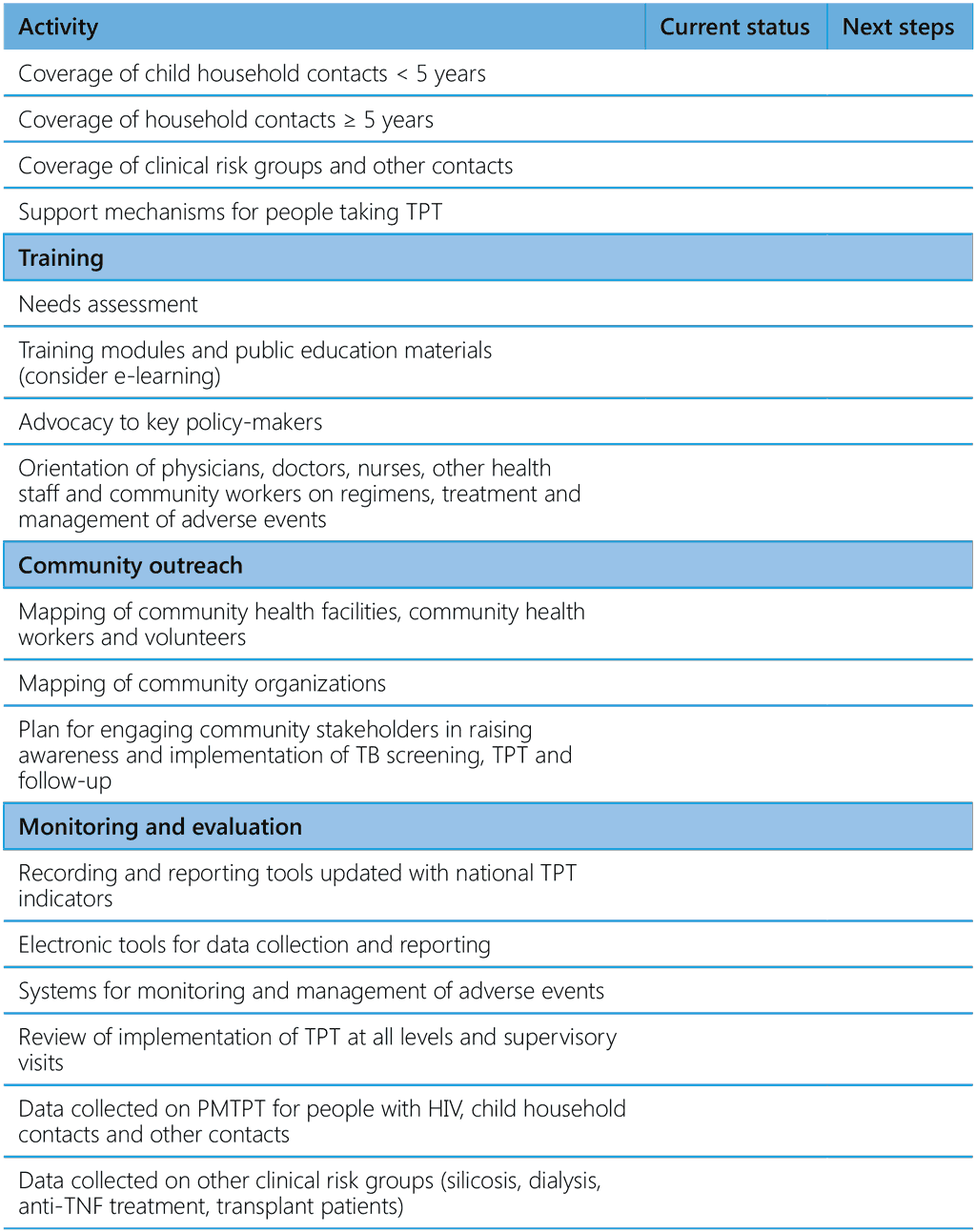
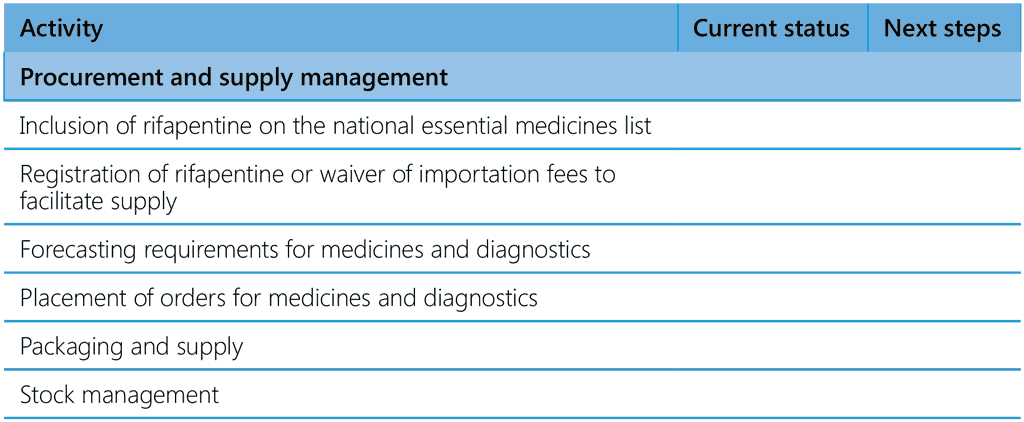
ART, antiretroviral therapy; CXR, chest radiography; HIV, human immunodeficiency virus; PMTPT, programmatic management of TB preventive treatment; TB, tuberculosis; TNF, tumour necrosis factor; TPT, TB preventive treatment; WHO, World Health Organization
References
- Guidance on conducting reviews of tuberculosis programmes. Geneva: World Health Organization; 2024 (https://www.who.int/publications/i/item/9789240085817).
- Guidance for national strategic planning for tuberculosis. Geneva: World Health Organization; 2022 (https://www.who.int/publications/i/item/9789240052055).
 Feedback
Feedback