Book traversal links for 2.4.2 LPAs
LPAs are a family of DNA strip-based tests that detect mutations associated with drug resistance. They do this either directly, through binding DNA amplification products (amplicons) to probes targeting the most commonly occurring mutations (MUT probes), or indirectly, inferred by the lack of binding the amplicons to the corresponding wild-type probes.
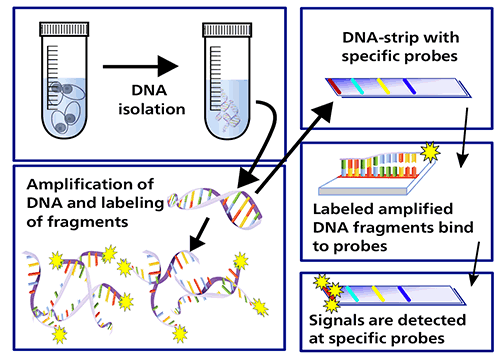
First-line LPAs
First-line LPAs (FL-LPAs) such as GenoType MTBDRplus and NTM+MDRTB Detection Kit allow the detection of resistance to RIF and INH. WHO recommends using FL-LPAs in the following situations (7):
- For people with a smear-positive sputum specimen or a cultured isolate of MTBC, commercial LPAs may be used as the initial test instead of phenotypic DST to detect resistance to RIF and INH.
Notes:
- These recommendations apply to the use of FL-LPAs for testing smear-positive sputum specimens (direct testing) and cultured isolates of MTBC (indirect testing) from both pulmonary and extrapulmonary sites.
- Conventional culture-based phenotypic DST for INH may still be used to evaluate a person with TB when the LPA result does not detect INH resistance. This is particularly important for populations with a high pretest probability of resistance to INH.
- FL-LPAs are not recommended for the direct testing of sputum-smear-negative specimens for the detection of MTBC.
Second-line LPAs
Second-line LPAs (SL-LPAs) such as the GenoType MTBDRsl test allow the detection of resistance to FQs and AMK. WHO recommends using SL-LPAs in the following situations (7):
- For a person with confirmed MDR/RR-TB, an SL-LPA may be used as the initial test, instead of phenotypic DST, to detect resistance to FQs and AMK.
Notes:
- This recommendation applies to the use of SL-LPA for testing sputum specimens, irrespective of the smear status, and cultured isolates of MTBC from both pulmonary and extrapulmonary sites.
- Culture-based phenotypic DST may be useful in evaluating people with negative SL-LPA results, particularly in populations with a high pretest probability for resistance to FQs or AMK.
- SL-LPA tests are also useful for detecting FQ resistance before starting therapy for Hr-TB.
 Feedback
Feedback