Book traversal links for 2. Identifying populations for TB preventive treatment


The first step in scaling up PMTPT is identification of populations eligible for TPT. The WHO guidelines on TPT recommend (13) that the target populations are those that fulfil one or more of the following criteria:
- high prevalence of TB infection;
- high risk of progression to TB disease; and
- higher incidence of TB disease than in the general population, indicating high TB transmission.
The benefits of TPT outweigh the potential risk of acquiring TB or drug toxicity. WHO thus recommends two broad groups at risk for systematic assessment of eligibility and provision of TPT:
- People at elevated risk of progression from infection to TB disease:
- people with HIV and
- patients with silicosis, starting or preparing for tumour-necrosis factor (TNF) treatment, receiving dialysis or preparing for an organ or haematological transplant.
- People at increased likelihood of exposure to TB disease:
- household contacts of people with bacteriologically confirmed TB, usually subdivided into:
- children < 5 years and
- children ≥ 5 years, adolescents and adults; and
- people who live or work in institutional or crowded settings, such as prisoners, health workers, recent immigrants from countries with a high TB burden, homeless people and people who use drugs.
- household contacts of people with bacteriologically confirmed TB, usually subdivided into:
2.1 Populations eligible for TPT
2.1.1 People with HIV
People with HIV are about 18 times more likely to develop TB disease than those without HIV infection and should be prioritized for systematic evaluation and TPT in all settings (9). Despite major progress in access to and the effectiveness of antiretroviral treatment (ART), TB is the most frequent cause of AIDS-related deaths worldwide (20). In 2022, TB caused over 167 000 deaths among people with HIV, representing about one fourth of all HIV deaths (9). TPT increases the survival of people with HIV even when they are on ART (21). TPT also provides additional protection when given immediately after successful completion of treatment for TB disease in people with HIV (21–23).
While TPT should be considered for infants aged < 12 months living with HIV who have a history of contact with a person with TB, children with HIV aged ≥ 12 months should be considered for TPT irrespective of contact with a person with TB. TPT is recommended for children with HIV, regardless of whether they are on ART or not. The evidence for an additive benefit of TPT among children with HIV on ART is limited, but it is plausible, given the efficacy observed among adults with HIV receiving ART plus TPT. Similarly, the effect of TPT in children with HIV after successful completion of TB treatment is largely extrapolated from benefits observed in adults exposed to reinfection and recurrence of TB.
Infants born to HIV-infected mothers are vulnerable to early TB infection due to the mother’s risk of contracting TB disease (24,25). Given the poor outcomes of TB disease in exposed infants, it is important to consider TPT for infants who show no signs of TB disease. Excluding TB disease before starting TPT in infants who are low birth weight or malnourished requires very careful evaluation. Prevention of mother-to-child transmission of HIV is an important platform for screening these infants for TB disease. A strong link should therefore be established between mother-and-child health services and national TB programmes (NTPs) (26).
WHO also recommends provision of TPT to children with HIV who successfully complete treatment for TB disease. People with HIV face a higher risk of recurrence of TB disease than HIV-negative individuals. While a complete course of TB treatment with a four-drug regimen has a very high success rate and a very low incidence (2–3%) of recurrence, the risk is several times higher in people with HIV, which can be due to treatment failure, emergence of drug resistance during therapy or reinfection with a new strain of M. tuberculosis (27–30). In a study of people with HIV whose initial episode of TB was considered to have been cured, 14% experienced a recurrence of TB, in nearly 90% of whom was due to reinfection with a different strain of M. tuberculosis (31). Interventions to minimize recurrence of TB include: ensuring completion of the initial course of TB treatment, effective infection control measures in clinics and communities frequented by people with HIV, and TPT in former TB patients who completed treatment successfully (secondary TPT) (32,33).
2.1.2 Household contacts (regardless of HIV status)
Household contacts of people with TB are well recognized as being at risk for TB infection and TB disease, including prevalent TB detected at the time of initial contact and incident TB occurring within the subsequent 2–5 years. The prevalence of TB infection among household contacts exceeds 50% in many low- and middle-income countries, while the prevalence of TB disease among household contacts in those countries was 3–5% (34,35). The results of the PHOENIx feasibility study in eight high TB burden countries showed that, of 1007 household contacts of 284 multidrug-resistant TB (MDR-TB) patients, 12% had TB disease and 72% were infected with TB (defined as either TST- or interferon-γ release assay (IGRA)-positivity) (36). Children < 5 years who are household contacts of people with TB are at significantly higher risk of acquiring TB infection and progressing rapidly to TB disease. A systematic review and meta-analysis were conducted to estimate the risk of children (defined as people aged < 19 years) for TB disease after close exposure and the effect on risk of TPT, bacille Calmette-Guérin (BCG) vaccination and time since TB exposure (37). It was found that the risk of exposed infants with evidence of TB infection for TB disease within 2 years of being evaluated as a contact and did not receive TPT was 18%. In contrast to previous estimates that suggested a lower risk for children aged 2–5 years, this study found that children in this age group had an equally high risk of developing TB within 2 years (19%). The effectiveness of TPT in preventing development of TB disease was estimated to be 91% for children and adolescents with TB infection. Another important finding was that 61% of children and adolescents and 83% of all children aged < 5 years with TB infection who developed TB disease did so within weeks of the initial contact investigation.
Children < 2 years of age are also at greater risk for severe and disseminated forms of TB and at very high risk of morbidity and mortality. Therefore, TPT is strongly recommended once TB disease has been ruled out. Screening of household contacts for TB disease is a high priority for all TB control programmes because it has a high yield and is a cost-effective active case-finding (ACF) strategy (38).
Household contact investigation ensures timely treatment for TB disease and TPT and thereby reduces transmission and improves TB-related outcomes. It is also an opportunity to improve infection control in a household and can result in important financial benefits for the entire family. The occurrence of TB in a family has serious social and economic effects, including catastrophic costs due to loss of income or the cost of health care. By investigating, detecting and treating both TB disease and TB infection, transmission in a household can be stopped and the catastrophic costs and dire health outcomes due to TB prevented. This holistic “family health” approach offers more efficient TB care to all members. Providing TPT to all family members at the same time and while the index TB patient is still receiving treatment and care can increase understanding of the impact of TPT and increase the cost– effectiveness ratio of interventions such as home visits. Nutritional assessment and support for contacts further protect them from progression of disease (see also section 7).
2.2 Integrating systematic screening for TB disease
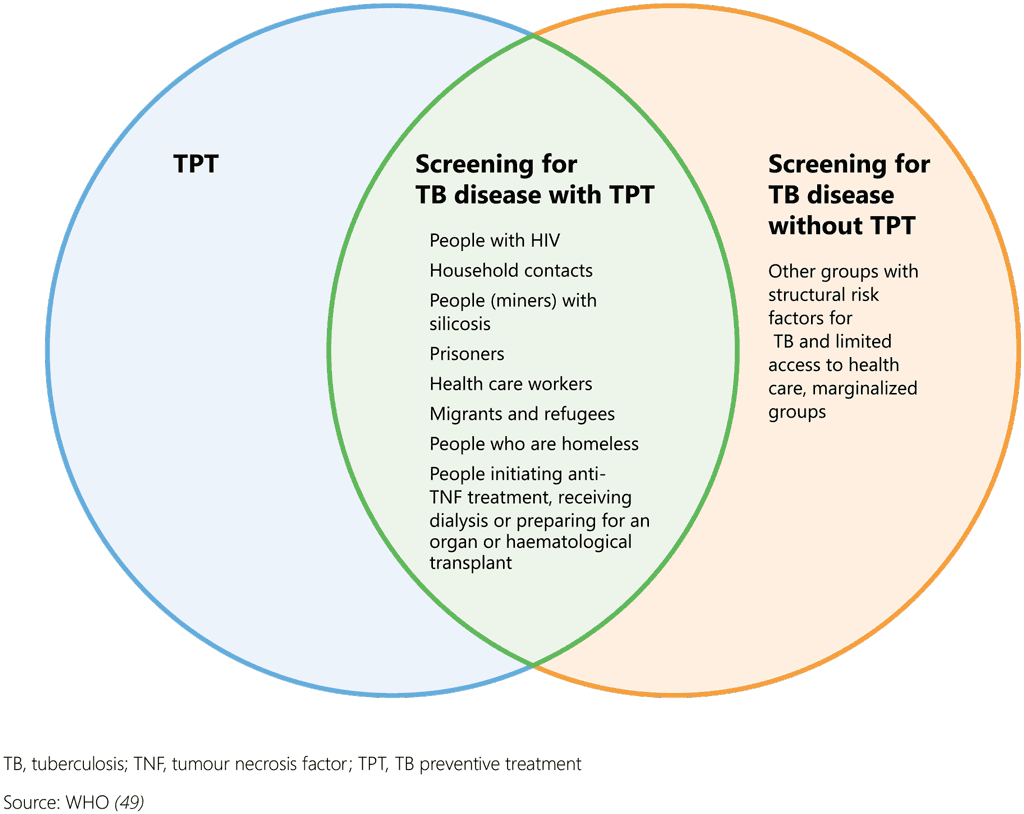
WHO recommends systematic screening for TB disease among at-risk populations that are also eligible for TPT (Fig. 3). These groups are people with HIV, household and close contacts of people with TB, miners exposed to silica dust and prisoners (strong recommendation for screening for TB disease). In addition, screening for TB disease is conditionally recommended for people with structural risk factors for TB and limited access to health care, such as urban poor, homeless people, refugees, migrants and other vulnerable or marginalized groups.
Integration of TPT services into systematic screening for TB disease in these populations allows NTPs to progress towards the UNHLM targets of reaching 90% of people with TB prevention and care, contributes to person-centred care and ensures efficient use of resources (13,17).
National programmes may integrate systematic evaluation for TPT eligibility in these populations, depending on individual risk in terms of recent exposure to a TB patient, immune status and other comorbidities, and provide access to TPT once TB disease has been ruled out. National programmes may adapt the algorithm for other risk groups (see Fig. 6 in section 4) when integrating ACF and TPT for populations at risk.
The scope of activities during contact investigations can be extended beyond TB services, depending on the country. For example, when the index TB patient is HIV positive, household contacts should systematically also be offered HIV counselling and testing, and, if malnutrition is noted during contact investigation, all contacts should be screened and assessed for nutritional factors.
Fig. 3. Populations for which PMTPT and screening for TB disease are recommended
2.3 National policy for identifying TPT target populations
A national policy for identifying TPT target populations should be established through the following steps.
- Establish a national technical working group or extend the mandate of an existing technical working group or equivalent to advise the ministry of health and the national TB and HIV programmes. The technical working group may consist of national experts, stakeholders in national TB, HIV, maternal and child health and other relevant programmes, representatives of patient groups, civil society, front-line health providers, national research institutes, technical partners and WHO.
- Review national policies and guidelines. The national technical working group may review current national policies and guidelines for PMTPT and lead updating and alignment with the latest global guidelines. The group may be mandated to lead identification of target populations and strategies for reaching those populations within PMTPT (Annex 3).
- Assess the situation. The technical working group and/or national programme may review the following to guide decisions on identifying target populations for PMTPT:
- the burden of TB disease (and TB infection) in various at-risk populations;
- capacity of the health system (staff, skills and equipment) to assess the intensity and risk of TB exposure and exclude TB disease;
- the availability of financial resources and identification of gaps in nationwide scaling up of TPT services and opportunities to mobilize additional resources; and
- opportunities to integrate PMTPT into systematic screening for TB disease.
- Other target populations for PMTPT may be identified by a small-scale demonstration or phased implementation of PMTPT among target populations to identify operational issues and inform strategies to reach them with TPT services. Extensive research to review the efficacy of the TPT regimen is not required, as the WHO guidelines are based on in-depth reviews of evidence from the latest clinical trials. Moreover, any country-specific or population-specific study might result in delay in scaling-up TPT services, thereby denying the benefits of TPT to vulnerable populations.
- As a minimum aim, all countries should achieve universal coverage of TPT for people with HIV and household contacts of TB patients aged < 5 years.
- Prioritize individuals who are likely to have acquired TB infection recently, such as children, recent immigrants from countries with a high TB burden, recent contact with a TB patient or documented conversion of a TB infection test from negative to positive.
Particularly for populations in congregate settings, surveillance and treatment of TB disease and infection control measures must be implemented effectively (13,39). These are essential prerequisites for deciding to provide TPT services to such populations. Without good measures for control of airborne infection, the sustained benefits of TPT may be compromised by a high risk for reinfection. Therefore, once target populations for TPT are identified, ministries of health, donors and stakeholders should support capacity-building in programmes to strengthen infection control and ensure access to rapid TB diagnosis and treatment.
2.4 Reaching TPT target populations
2.4.1 Reaching people with HIV and other target populations
All HIV testing and treatment facilities, including community HIV care and support, should systematically intensify TB case finding, TPT and TB infection control. These require good collaboration between national TB and HIV programmes (40).
All people with HIV should be screened for TB symptoms at every opportunity or contact with a health worker. CXR screening increases sensitivity for detecting TB disease in people with HIV in regular care, and annual screening with CXR might be a good strategy for finding additional people with TB in this population (17). People with TB symptoms or TB-related abnormalities on CXR should be referred for diagnostic testing, and those without symptoms and a normal CXR should be evaluated for eligibility and started on TPT, as appropriate.
Adults and adolescents with newly diagnosed HIV should always be screened for TB. CRP is more specific than symptom screening alone and could be considered an additional screening test for this group (17). People with TB symptoms or CRP values > 5 mg/L should be referred for diagnostic testing, and those without symptoms or CRP values of ≤ 5 mg/L should be evaluated for eligibility and started on TPT, as appropriate.
National TB and HIV programmes should provide resources and monitor sites to ensure implementation and quality improvement measures when gaps are noted (such as lack of screening, cursory screening, lack of linkage to TPT).
For other target populations, the national programme should tailor TB screening and TPT services to the needs and capacities of the existing health infrastructure. The approach should optimize and synergize delivery of TPT services with other health and social welfare services. Target populations and screening approaches should be monitored regularly to ensure their efficiency and to improve services.
2.4.2 Strengthening investigation of household contacts
Contact investigation is an important first step for both ACF and TPT. It consists of systematic identification of people with TB disease and contacts of index TB patients who should receive TPT. After identification, clinical evaluation and/or testing and provision of access to appropriate TB treatment should be provided for people with confirmed TB or TPT for those without TB disease. It should be a standard component of all national TB control programmes. Moreover, contact investigation is good public health practice and is essential for tracking several infectious diseases (such as coronavirus disease 2019). Therefore, ministries of health should invest in strengthening health system capacity. If mechanisms for contact investigation are in place, national programmes should strengthen them to ensure that all contacts aged ≥ 5 years are also covered. If such a mechanism is lacking, the ministry of health should dedicate the necessary human and financial resources to establish effective mechanisms for contact investigation. An indicative list of items to be considered for determining appropriate unit costs for budgeting and planning to strengthen contact investigations is provided in Annex 4. In addition to extra funding, NTPs should ensure effective contact investigation (Fig. 4)
Fig. 4. Building blocks for strong contact investigation
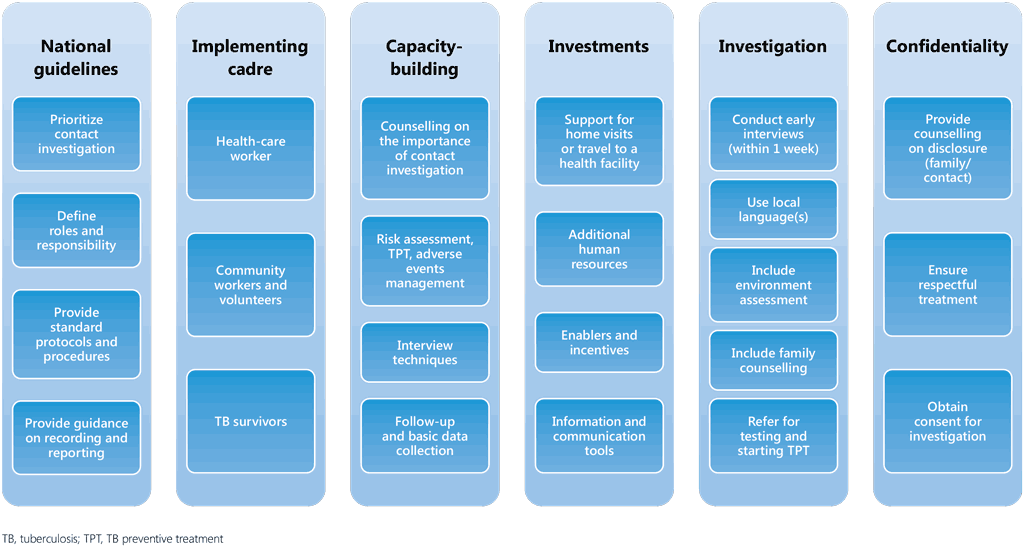
To ensure efficient contact investigation, the following elements should be included.
- Provide national guidelines that:
- define priority populations for contact investigation (household and beyond);
- define the model of care, facility, community or a combination;
- define the roles and responsibilities of programme personnel, health-care workers and community health workers in reaching contacts, screening for TB disease by symptoms and other tests such as CRP and CXR, and referral for clinical evaluation and diagnostic testing; include responsibilities in job descriptions;
- provide standard guidance and approaches for reaching contacts, and conduct investigations to ensure uniform implementation;
- define the data elements to be captured in the index patient’s record and/or digital tools for contact investigations (see also section 7);
- in a community model, include tools or referral slips for recording data on TB screening and referral of identified contacts; and
- define the level of service delivery points for systematic recording and reporting and frequency; and
- provide locally tested messages to generate demand and to educate patients.
- Use human resources and mechanisms in other disease programmes (such as the public health response model promoted by the US President’s Emergency Plan for AIDS Relief for people with HIV) for contact investigation, and ensure sustainability and efficiency. TB and HIV screening could be integrated.
- Train health workers and community workers in conducting a contact investigation, including the following (41):
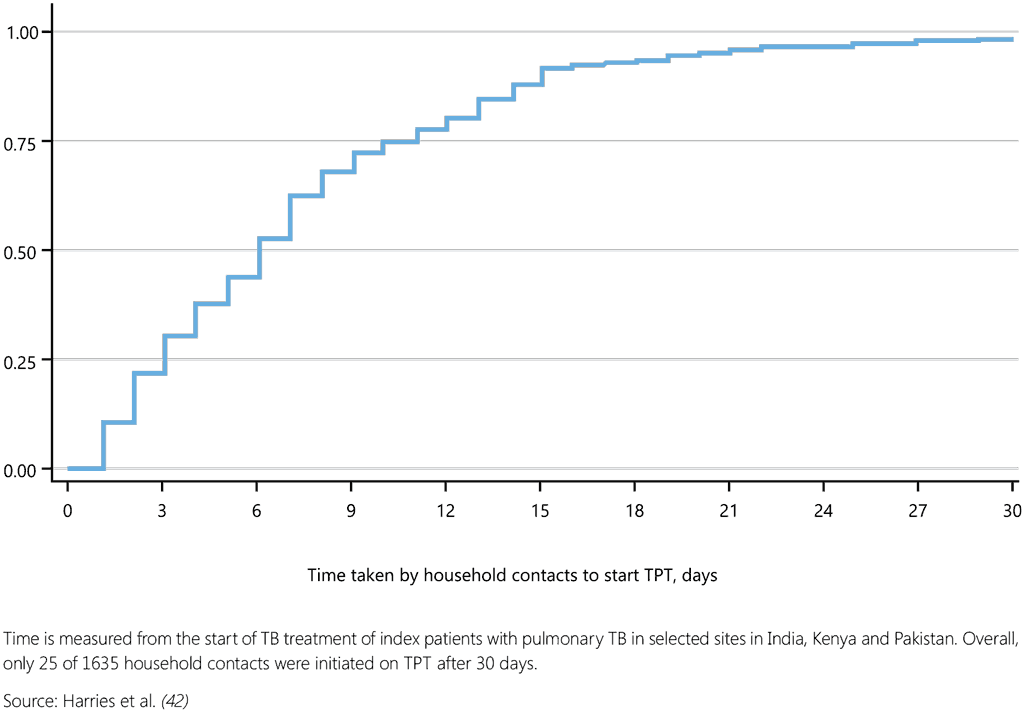
- The index person with TB should be interviewed as soon as possible after diagnosis, preferably within 7 days, to elicit details about household and other close contacts (see Box 1 for information on measuring timeliness) (42). Health providers should clearly and sensitively explain the urgency of initiating contact investigations to the index patient in view of the increased risk of progression to TB disease after recent exposure. A second interview may be required to elicit additional contacts and any missing information.
- While the focus of the contact investigation should be household members, additional contacts should be considered as per the national guideline for evaluation, including contacts at the workplace, residential care facilities, residential schools, long-term care facilities, prisons, other correctional facilities and acute medical care facilities. This is especially important when exposure is likely to have been prolonged and the index person is likely to be highly infectious (e.g. with prolonged cough or extensive cavitary disease on CXR).
- Ideally, interviews should be conducted by a person who speaks the same language as the index person and is familiar with his or her social and cultural context.
- Education of the index person and household members on the benefits of taking TPT and the risks of not taking it should be central to contact investigation. The aim should be to enable an informed decision to receive a complete course of a TPT regimen.
- Counselling of index people should also help them to appreciate the importance of identifying all close contacts. This will make it possible to reach more people at risk.
- In the community model, approval should be sought from the index person for a home visit. In addition to counselling the index person, arrangements should be made to counsel contacts before starting TPT.
- Preferably, health-care workers who conduct contact investigations should visit the home or workplace of the index person, conduct interviews and emphasize the importance of identifying and evaluating contacts, conduct screening for TB disease, collect accurate information about the intensity and duration of exposure, and ensure that all relevant contacts are referred for further evaluation and treatment decisions (Boxes 2 and 3) (43). Information to be documented during a contact investigation includes the name of the person who identified the contact, the person who identified the index person with TB, the demographics of the contact, the results of TB screening tests and tests for TB infection (if done), the date on which eligibility for TPT was determined and the decision to prescribe TPT. Visits might have to be made outside normal working hours, as contacts may be at work or school during those hours.
- Home visits also provide an opportunity to identify any requirement for social or nutrition support or education on infection control measures, and the health provider may subsequently link the index person and contacts to relevant social and nutritional support programmes. During the home visit, the health provider should assess the residence and provide counselling and education to family members on symptoms of TB. A recent cluster-randomized, controlled trial in India showed that provision of nutritional supplementation to households of people with microbiologically confirmed pulmonary TB prevented further TB cases among contacts (44). (Nutritional interventions for TB prevention are to be evaluated by a WHO Guideline Development Group on undernutrition (45).) If required, prompt medical attention and referrals should be made, especially for child contacts and people with HIV, in whom TB can progress rapidly. HIV testing and counselling should also be offered, including to the biological children of any adults with HIV.
- If the home or workplace cannot be visited, the index person may be interviewed at a health facility and contacts listed. The complete address and modality for future communication should be mutually agreed with the index person (such as phone numbers, email, contact of an intermediary or treatment provider). Responsible people or health-care workers should then systematically follow up the index person.
- After contact investigation, health-care workers should refer relevant contacts to a health facility for symptom screening, possible screening with CXR, diagnostic testing for TB and TB infection when indicated, and evaluation for eligibility for TPT.
- Information from the interviews should be recorded maintaining confidentiality.
- Budget and cost implementation of contact investigation. (See Annex 4 for further details.)
- Ensure that all enablers and incentives are costed, including travel costs.
- Health workers who conduct home visits and/or families might require support for travel to a facility; the costs should not be borne by affected families.
- Information and communication tools should also be costed.
- Provide guidance on M&E (see also section 8):
- Use standard tools and a protocol for data collection for contact investigation, data entry and analysis.
- Monitor the yield of contact investigations and the proportions of TB disease and TB infection detected.
- Provide guidance on confidentiality and informed consent (see also section 9):
- Maintaining confidentiality during contact investigation may be difficult because of social connections between index people and their contacts. All people should be treated with respect and confidentiality maintained. National programme guidelines on data protection, confidentiality and consent should be adhered to.
- When the index person is reluctant to provide information on household and social contacts, counselling should continue to gain the trust of the person with TB. The index person should not be coerced, nor should her/his TB treatment or services be made conditional on cooperation with contact investigation.
Annex 6 provides an example of data collection form for use during household contact evaluation.

Fig. 5. Time to initiation of TPT in household contacts

Box 3 provides an example of contact investigation by nurse-led outreach teams in Mozambique.

 Feedback
Feedback