Book traversal links for Annex 2. Supplementary table
Summary of changes to recommendations as included in the second edition of the Guidance for national tuberculosis programmes on the management of tuberculosis in children, 2014
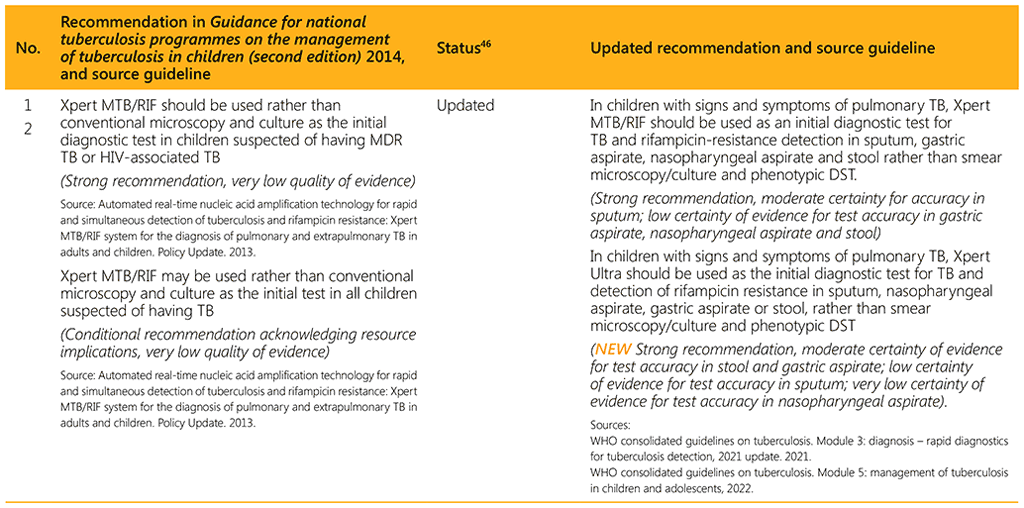

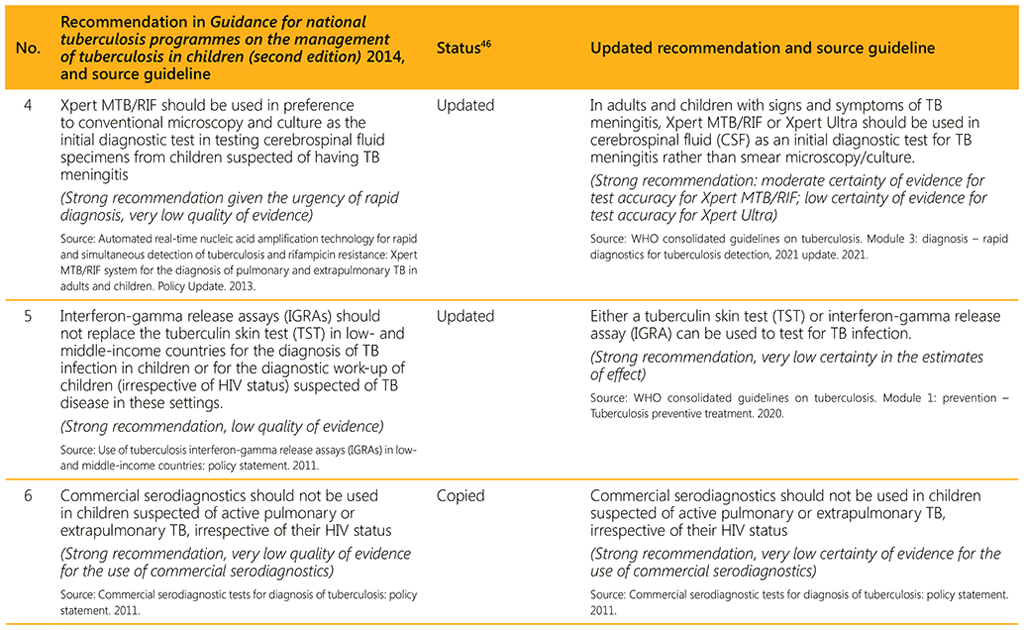
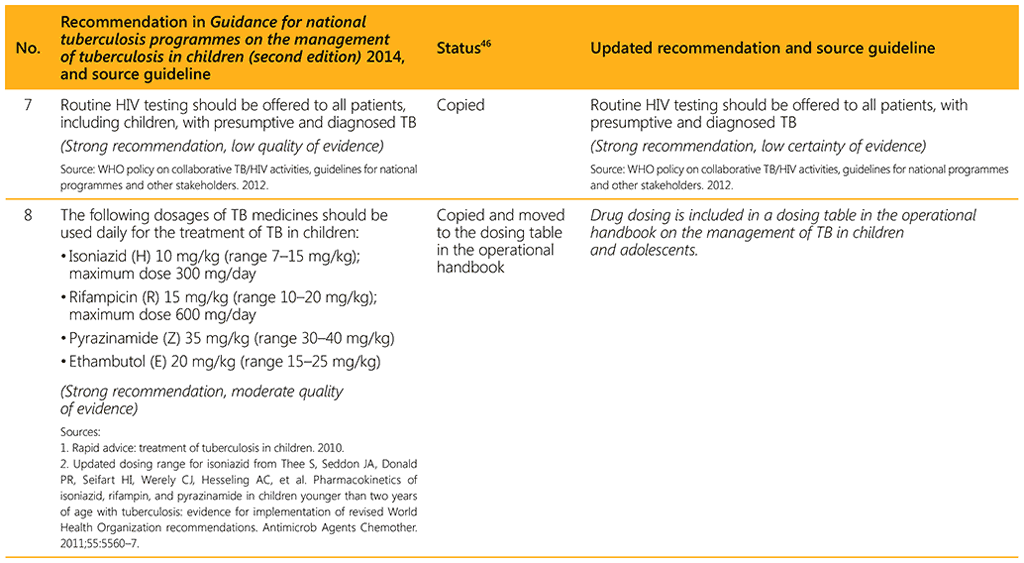
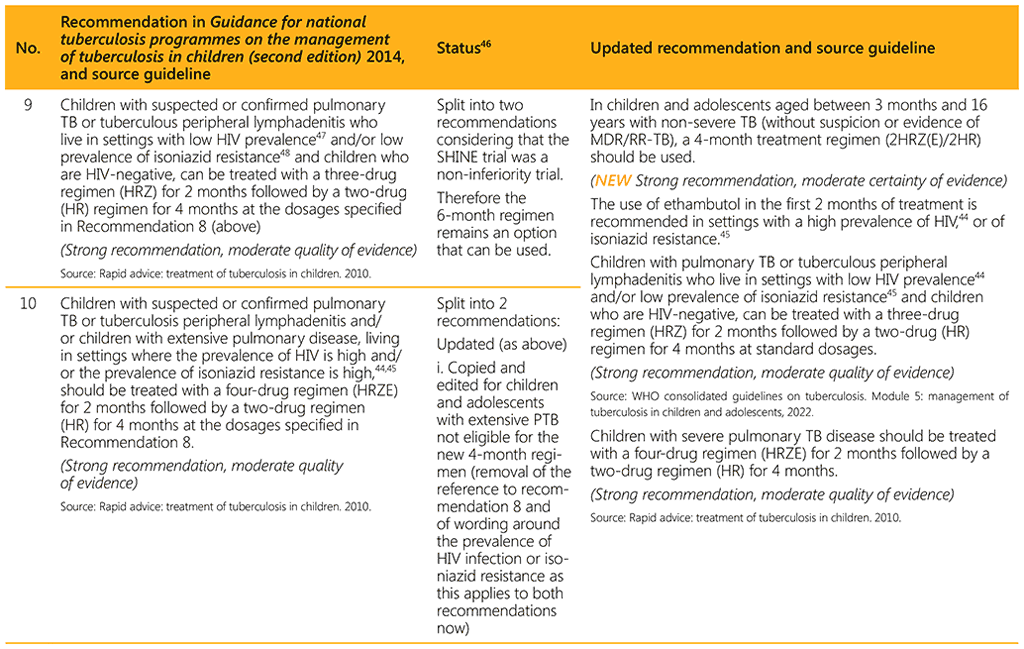
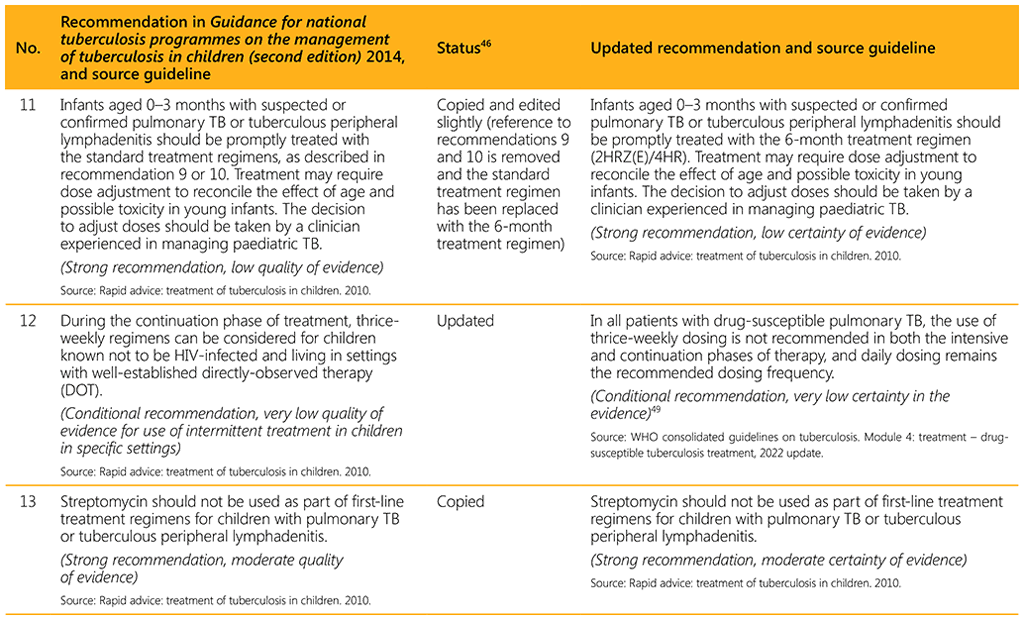
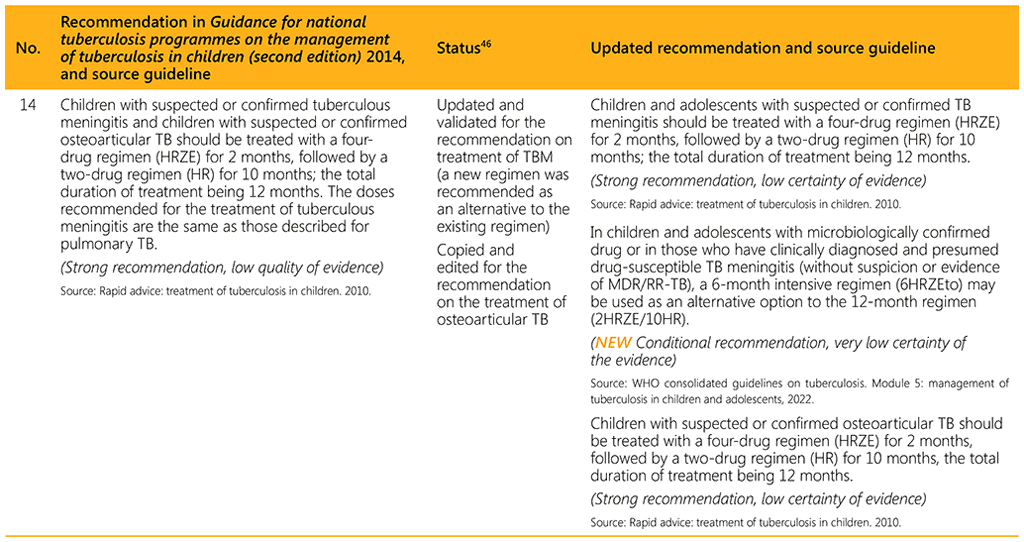
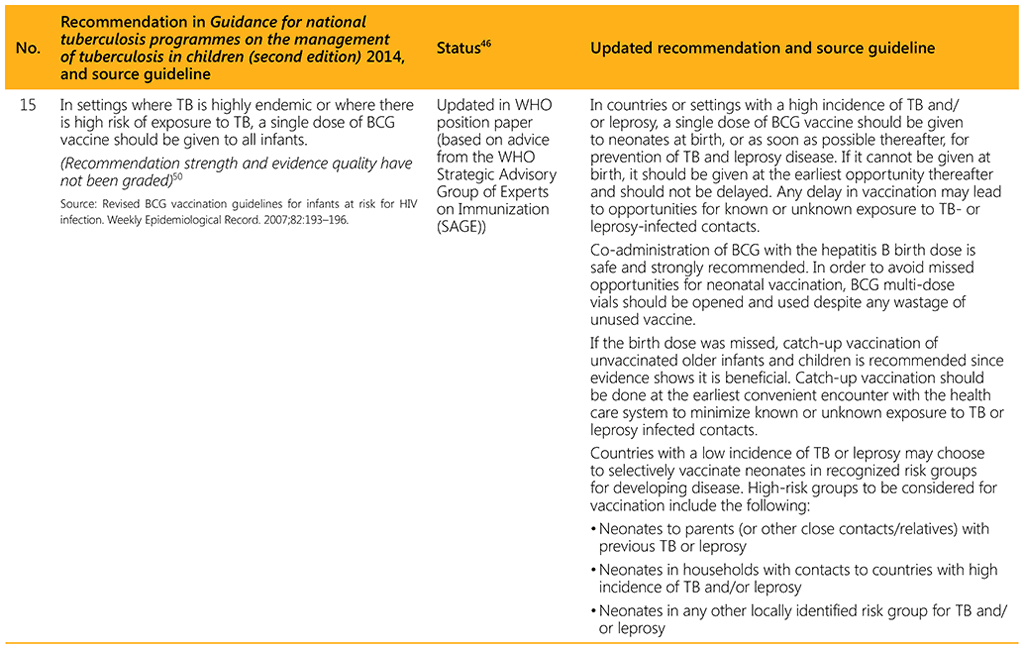
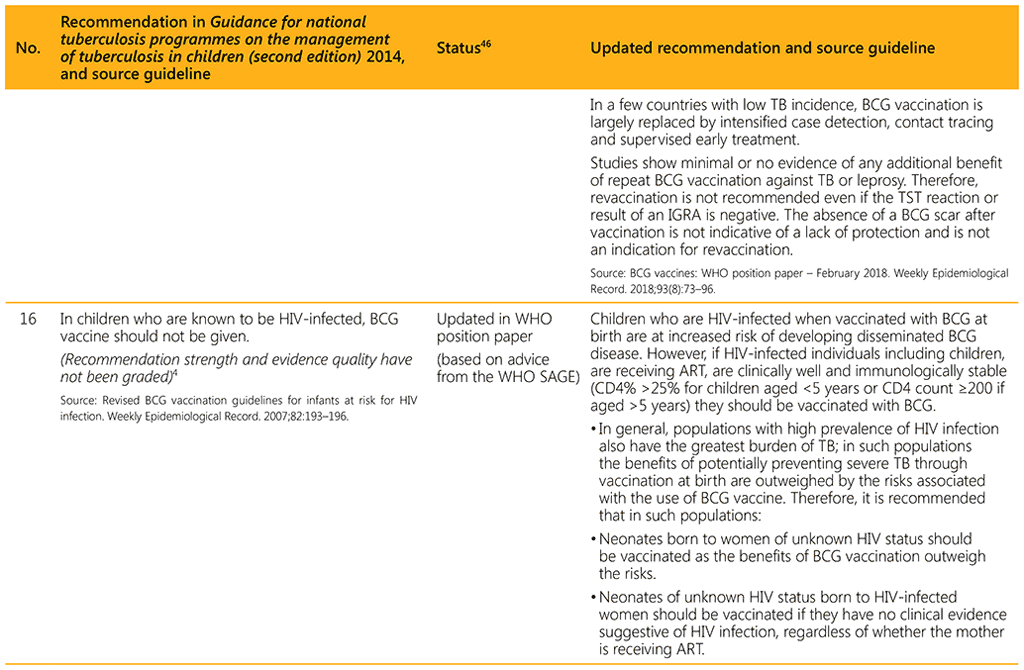
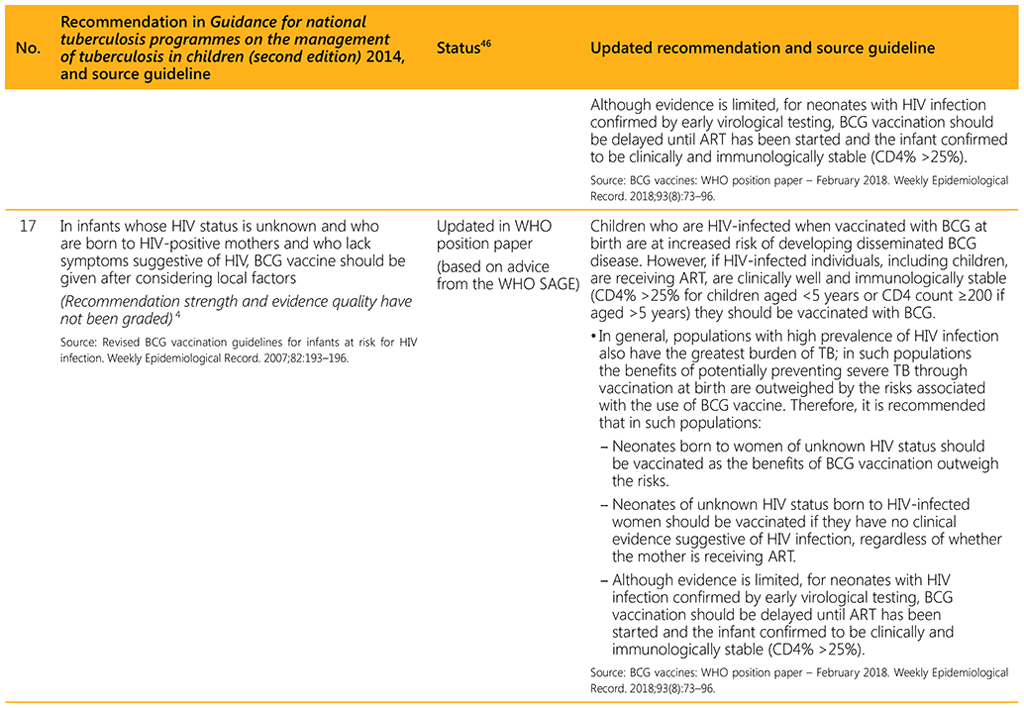
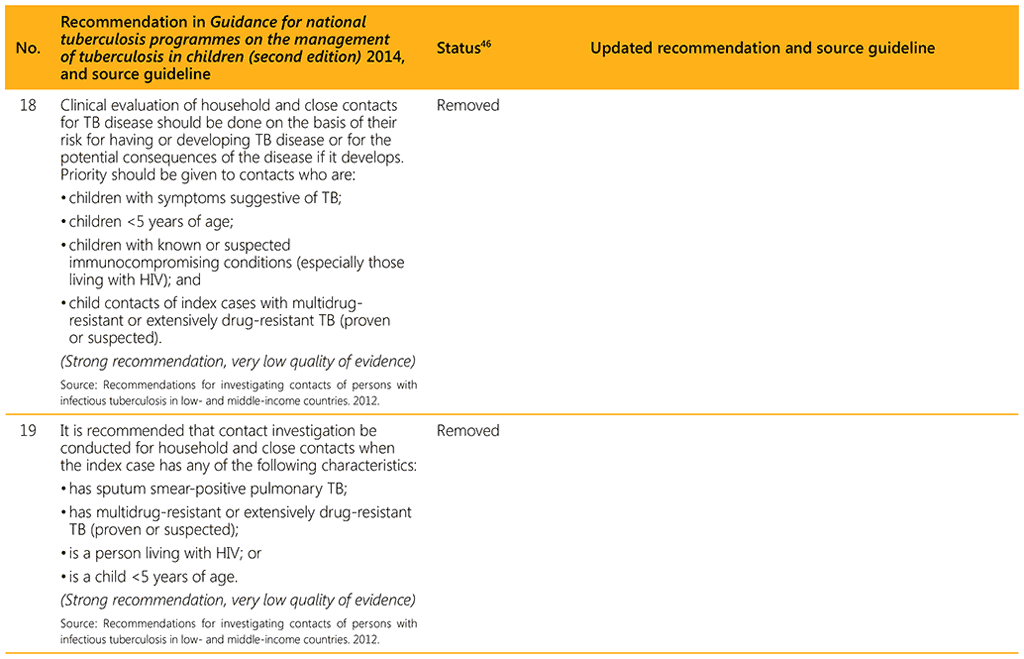
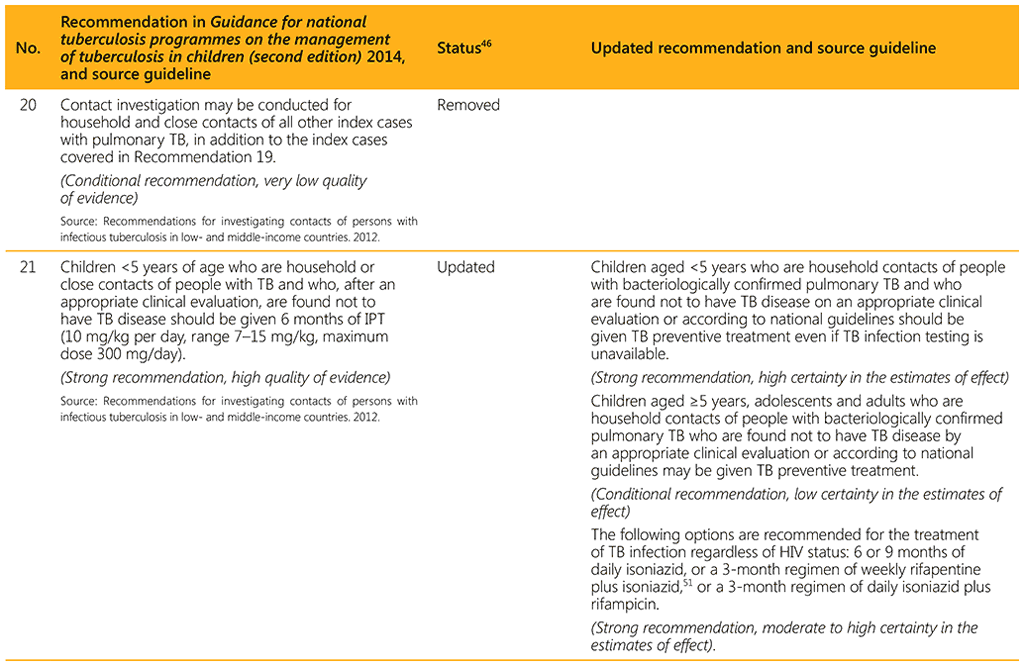
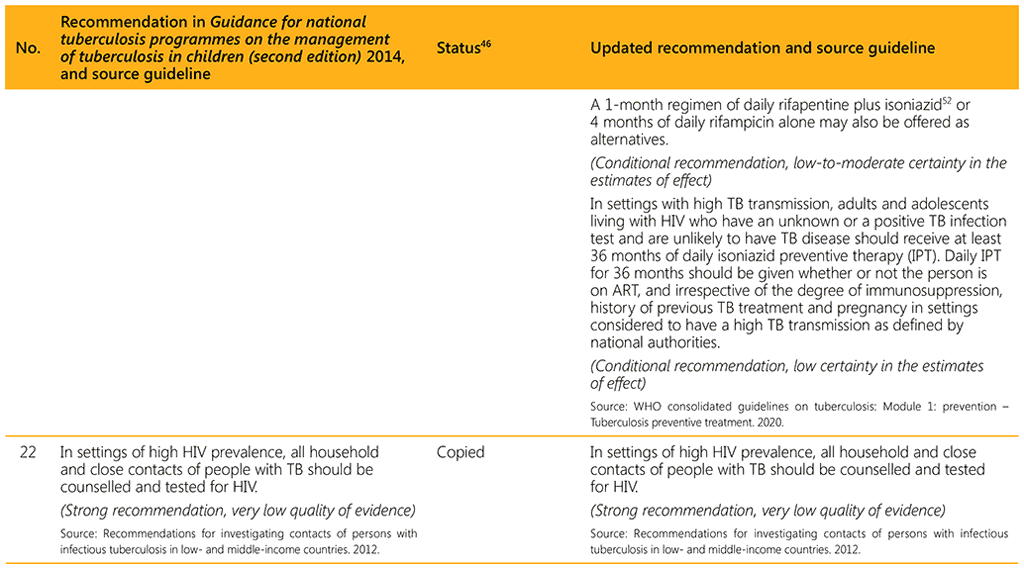
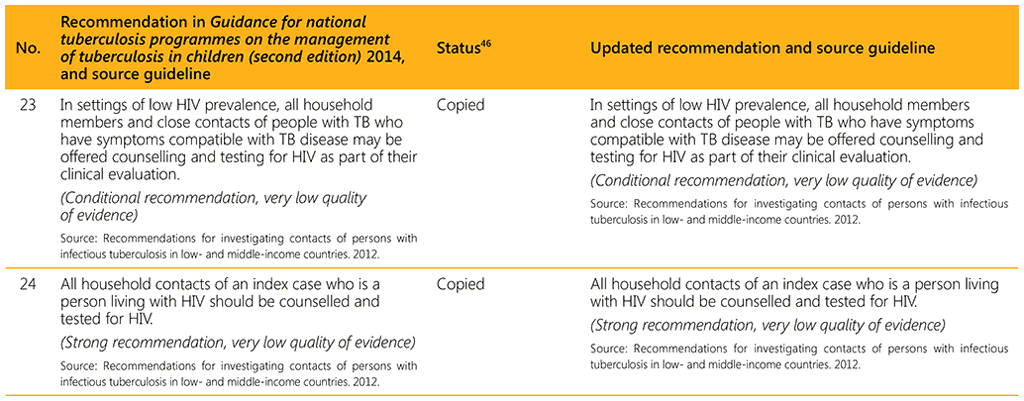
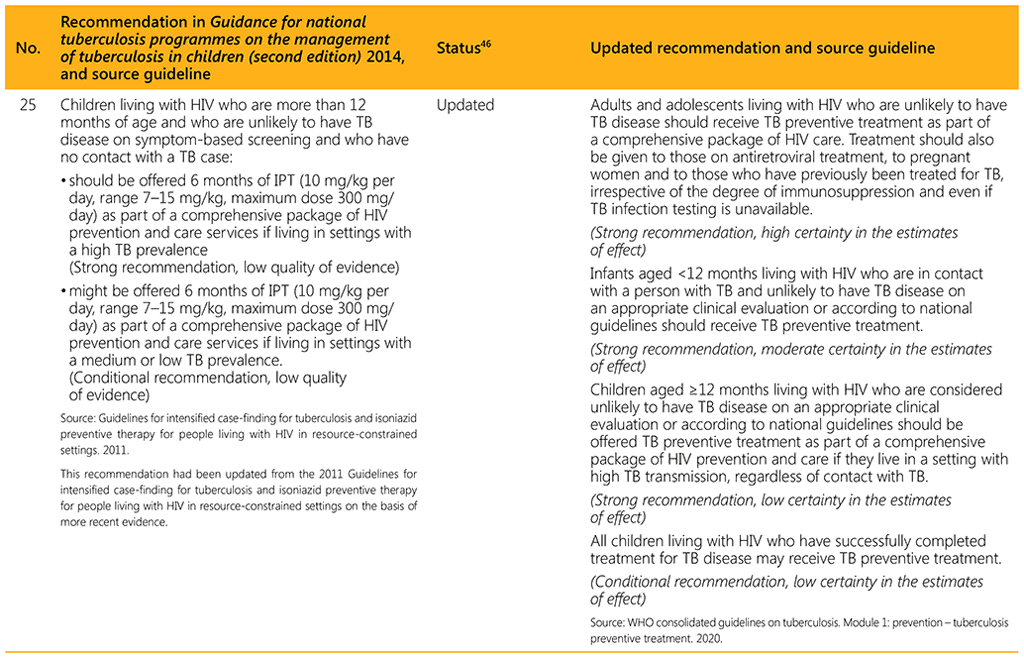
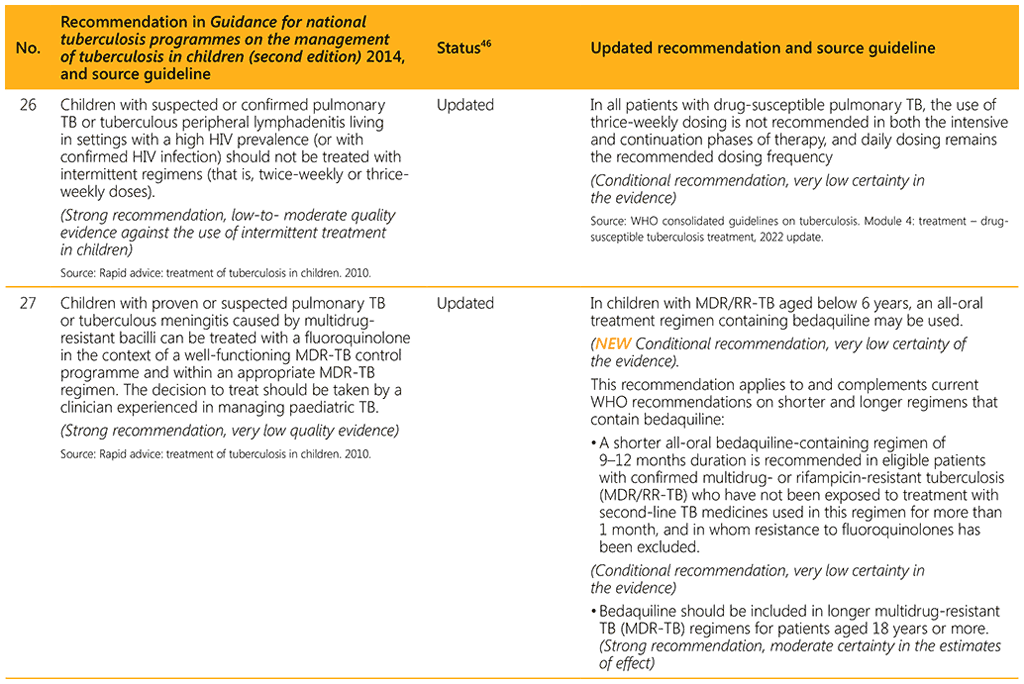

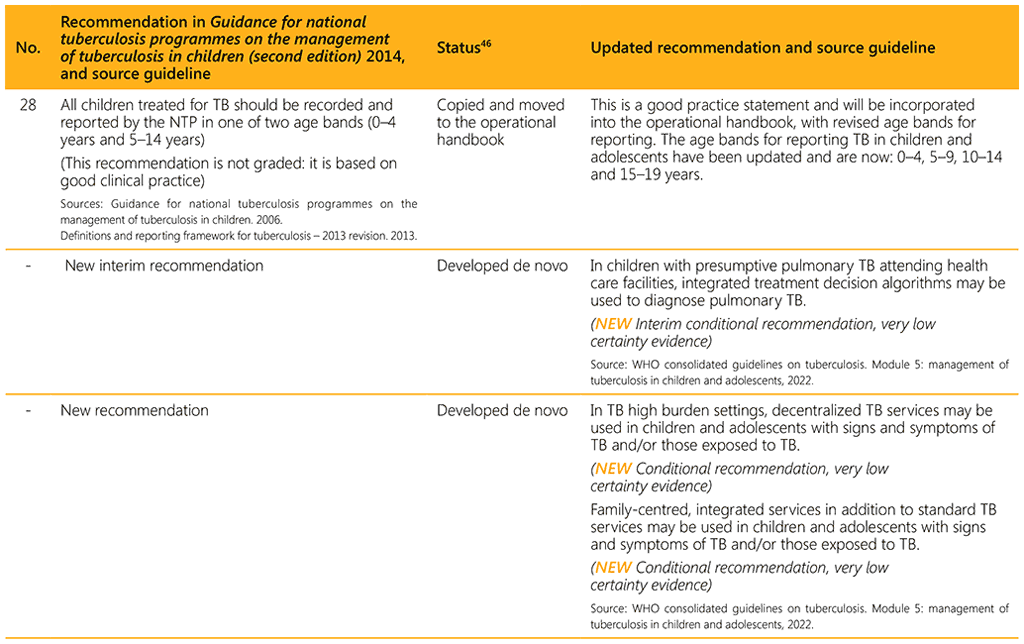
⁴⁶ Status: Removed (the recommendation has been superseded and is no longer relevant); Copied (the recommendation remains valid and is retained unchanged); Edited (There is no change in the evidence or in the intention of the recommendation, but the precise wording has been edited); Updated (a new evidence synthesis was conducted with review by the GDG with a full evidence-to-decision procedure); Developed de novo (a new topic, subgroup or intervention has been covered with a new evidence synthesis and a full evidence-to-decision procedure by the GDG)
⁴⁷ Defined as countries, subnational administrative units, or selected facilities, where the HIV prevalence among adult pregnant women is ≥1% or among TB patients is ≥5% in the 2014 Guidance for national tuberculosis programmes on the management of tuberculosis in children (second edition) (8).
⁴⁸ WHO does not intend to establish thresholds for low, moderate or high levels of prevalence of isoniazid resistance: NTPs will establish definitions for their own countries.
⁴⁹ Twice-weekly dosing is totally not recommended. See Guidelines for treatment of tuberculosis, fourth edition. Geneva: World Health Organization; 2010 (http://www.who.int/tb/publications/2010/9789241547833/ en/).
⁵⁰ The Global Advisory Committee on Vaccine Safety (GACVS) does not use the GRADE methodology for evaluating the quality of evidence; the BCG-related recommendations will therefore remain ungraded.
⁵¹ In ages two years and above.
⁵² In ages 13 and above.
 Feedback
Feedback